SUMMARY: Sickle Cell Disease or Sickle Cell anemia is an Autosomal Recessive disorder caused by mutations in the hemoglobin beta-globin gene, and affects approximately 100,000 Americans. It is estimated that it affects 1 out of every 365 African-American births and 1 out of every 16,300 Hispanic-American births. The average life expectancy for patients with Sickle Cell Disease in the US is approximately 40-60 years.
HbSS disease or Sickle Cell anemia is the most common Sickle Cell Disease genotype and is associated with the most severe manifestations. HbSS disease is caused by a mutation substituting thymine for adenine in the sixth codon of the beta-globin chain gene. This in turn affects the hemoglobin’s ability to carry oxygen and causes it to polymerize. This results in decreased solubility thereby distorting the shape of the red blood cells, increasing their rigidity and resulting in red blood cells that are sickle shaped rather than biconcave. These sickle shaped red blood cells limit oxygen delivery to the tissues by restricting the flow in blood vessels, leading to severe pain and organ damage (Vaso-Occlusive Crises). Oxidative stress is an important contributing factor to hemoglobin polymerization with polymer formation occurring only in the deoxy state. HbS/b-0 Thalassemia (double heterozygote for HbS and b-0 Thalassemia) is clinically indistinguishable from HbSS disease. Management of Sickle Cell Disease includes pain control, transfusion support and Hydroxyurea. None of the presently available therapies addresses the underlying cause of this disease nor do they fully ameliorate disease manifestations. Allogeneic bone marrow transplantation can cure this genetic disorder, but less than 20% of eligible patients have a related HLA-matched donor. There is therefore a great unmet need to find new therapies for Sickle Cell Disease.
Fetal hemoglobin which consists of two alpha and two gamma chains is produced in utero, but the level of gamma-globulin decreases postnatally as the production of beta-globin and adult hemoglobin, which consists of two alpha and two beta chains, increases. It has been noted that elevated levels of fetal hemoglobin facilitates oxygen delivery, prevents the sickling of red blood cells, and is associated with decreased morbidity and mortality in patients with Sickle Cell Disease. BCL11A gene is a repressor of gamma-globin expression, and fetal hemoglobin production in adult red blood cells. Downregulating BCL11A can therefore reactivate gamma-globin expression and increase fetal hemoglobin in RBC.
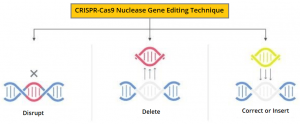
The Clustered Regularly Interspaced Short Palindromic Repeats (CRISPR)-Cas9 nuclease gene editing technique can be directed to cut DNA in targeted areas, enabling the ability to accurately edit (remove, add, or replace) DNA where it was cut. The modified hematopoietic stem cells are transplanted back into the patient where they engraft within the bone marrow and increase the production of fetal hemoglobin.
The researchers in this study used this gene-editing technique in Hematopoietic Stem and Progenitor Cells at the erythroid-specific enhancer region of BCL11A to down-regulate BCL11A expression in erythroid-lineage cells, restore gamma-globin synthesis, and reactivate production of fetal hemoglobin. CASGEVY®: (Exagamglogene Autotemcel) is the first cell-based gene therapy for the treatment of Sickle Cell Disease in patients 12 years and older. This product is made from the patients own hematopoietic stem cells, which are collected and modified, and are given back as a one-time, single-dose infusion as part of a Hematopoietic Stem Cell Transplant. Prior to this infusion, patient must undergo myeloablative conditioning (high-dose chemotherapy), a process that removes cells from the bone marrow, so they can be replaced with the modified cells.
CASGEVY® is the first FDA-approved non-viral cell therapy to utilize CRISPR/Cas9 genome editing technology, to modify patients (autologous) CD34+ Hematopoietic Stem and Progenitor Cells (HSPCs) at the erythroid-specific enhancer region of BCL11A. The FDA approval of CASGEVY® in December 2023 is based on a open-label, single-group, multi-center Phase 3 trial, involving adult and adolescent patients with Sickle Cell Disease. The trial focused on individuals with a history of at least two protocol-defined severe Vaso-Occlusive Crises (VOC) during each of the two years prior to screening.
This study included 44 patients, and their CD34+ HSPCs were edited with the use of CRISPR-Cas9. Patients underwent myeloablative conditioning with pharmacokinetically dose-adjusted Busulfan before the CASGEVY® infusion. The Primary end point was freedom from severe VOC for at least 12 consecutive months. A key Secondary end point was freedom from inpatient hospitalization for severe VOC for at least 12 consecutive months. Additionally, the safety of CASGEVY® was also assessed. The median follow-up was 19.3 months.
Out of the 44 patients treated with CASGEVY®, 30 patients had sufficient follow-up time, to be evaluated. Notably, all treated patients achieved successful engraftment, a crucial aspect confirming the efficacy of the CRISPR/Cas9 genome editing technology in modifying hematopoietic stem cells, and no instances of graft failure or rejection were reported, affirming the safety and viability of CASGEVY® as a therapeutic option.
This study met the Primary endpoint, and it was noted that 97% (29 patients) were free from VOC for at least 12 consecutive months and 100% (all 30 patients) were free from hospitalizations for VOC for at least 12 consecutive months (P<0.001 for both comparisons against the null hypothesis of a 50% response). Patients were free from VOC for a mean duration of 22.4 months. Early and sustained increases in total and fetal hemoglobin levels was noted, with a total hemoglobin level of 10.4 to 13.4 gm/dL at month 3, 10.7 to 14.3 gm/dL at month 6, and normal or near-normal levels (12.1 to 17.2 gm/dL) maintained thereafter. Improvements were also seen in all markers of hemolysis, including normalization of LDH and detectable haptoglobin levels, suggesting resolution of intravascular hemolysis. The safety profile of this intervention was generally consistent with that of myeloablative conditioning with Busulfan and autologous HSPC transplantation. No cancers occurred.
It was concluded that one-time treatment with non-viral ex-vivo CRISPR-Cas9 editing of the erythroid-specific enhancer region of BCL11A reactivated fetal hemoglobin production in erythrocytes, eliminating vaso-occlusive crises in 97% of patients with sickle cell disease for a period of 12 months or more. This high success rate underscores the therapeutic potential of CASGEVY® in mitigating the recurrent and debilitating crises associated with Sickle Cell Disease.
Exagamglogene Autotemcel for Severe Sickle Cell Disease. Frangoul H, Locatelli F, Sharma A, et al. for the CLIMB SCD-121 Study Group. N Engl J Med 2024;390:1649-1662.