SUMMARY: Prostate cancer is the most common cancer in American men with the exclusion of skin cancer, and 1 in 9 men will be diagnosed with Prostate cancer during their lifetime. It is estimated that in the United States, about 191,930 new cases of Prostate cancer will be diagnosed in 2020 and 33,330 men will die of the disease.
The development and progression of Prostate cancer is driven by androgens. Androgen Deprivation Therapy (ADT) or testosterone suppression has therefore been the cornerstone of treatment of advanced Prostate cancer and is the first treatment intervention. Androgen Deprivation Therapies have included bilateral orchiectomy or Gonadotropin Releasing Hormone (GnRH) analogues, with or without first generation Androgen Receptor (AR) inhibitors such as CASODEX® (Bicalutamide), NILANDRON® (Nilutamide) and EULEXIN® (Flutamide) or with second-generation, anti-androgen agents, which include, ZYTIGA® (Abiraterone), XTANDI® (Enzalutamide) and ERLEADA® (Apalutamide). Approximately 10-20% of patients with advanced Prostate cancer will progress to Castration Resistant Prostate Cancer (CRPC) within five years during ADT, and over 80% of these patients will have metastatic disease at the time of CRPC diagnosis. The estimated mean survival of patients with CRPC is 9-36 months, and there is therefore an unmet need for new effective therapies.
DNA damage is a common occurrence in daily life by UV light, ionizing radiation, replication errors, chemical agents, etc. This can result in single and double strand breaks in the DNA structure which must be repaired for cell survival. The two vital pathways for DNA repair in a normal cell are BRCA1/BRCA2 and PARP. BRCA1 and BRCA2 genes recognize and repair double strand DNA breaks via Homologous Recombination Repair (HRR) pathway. Homologous Recombination is a type of genetic recombination, and is a DNA repair pathway utilized by cells to accurately repair DNA double-stranded breaks during the S and G2 phases of the cell cycle, and thereby maintain genomic integrity. Homologous Recombination Deficiency (HRD) is noted following mutation of genes involved in HR repair pathway. At least 15 genes are involved in the Homologous Recombination Repair (HRR) pathway including BRCA1, BRCA2 and ATM genes. The BRCA1 gene is located on the long (q) arm of chromosome 17 whereas BRCA2 is located on the long arm of chromosome 13. BRCA1 and BRCA2 are tumor suppressor genes and functional BRCA proteins repair damaged DNA, and play an important role in maintaining cellular genetic integrity. They regulate cell growth and prevent abnormal cell division and development of malignancy.
Recently published data has shown that deleterious Germline and/or Somatic mutations in BRCA1, BRCA2, ATM, or other Homologous Recombination DNA-repair genes, are present in about 25% of patients with advanced prostate cancer, including mCRPC. Approximately 12% of men with mCRPC harbor a deleterious BRCA1 or BRCA2 mutation (BRCA1, 2%; BRCA2, 10%). Mutations in BRCA1 and BRCA2 also account for about 20-25% of hereditary breast cancers, about 5-10% of all breast cancers, and 15% of ovarian cancers. BRCA mutations can either be inherited (Germline) and present in all individual cells or can be acquired and occur exclusively in the tumor cells (Somatic). Somatic mutations account for a significant portion of overall BRCA1 and BRCA2 aberrations. Loss of BRCA function due to frequent somatic aberrations likely deregulates HR pathway, and other pathways then come in to play, which are less precise and error prone, resulting in the accumulation of additional mutations and chromosomal instability in the cell, with subsequent malignant transformation. HRD therefore indicates an important loss of DNA repair function. The PARP (Poly ADP Ribose Polymerase), family of enzymes include, PARP1and PARP2, and is a related enzymatic pathway that repairs single strand breaks in DNA. In a BRCA mutant, the cancer cell relies solely on PARP pathway for DNA repair to survive. PARP inhibitors trap PARP onto DNA at sites of single-strand breaks, preventing their repair and generating double-strand breaks that cannot be repaired accurately in tumors harboring defects in Homologous Recombination Repair pathway genes, such as BRCA1 or BRCA2 mutations, and this leads to cumulative DNA damage and tumor cell death.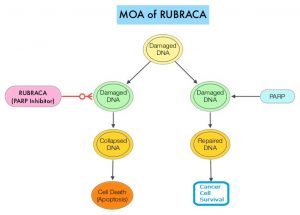
RUBRACA® (Rucaparib) is an oral, small molecule inhibitor of PARP. TRITON2 is an international, multicenter, open-label, single arm, Phase II trial, in which patients with BRCA-mutated mCRPC, who had progressed after one to two lines of next-generation Androgen Receptor-directed therapy and one taxane-based chemotherapy for mCRPC were included. In this study, 115 mCRPC patients with either Germline or Somatic BRCA mutations, with or without measurable disease were enrolled, of whom 62 patients (54%) had measurable disease at baseline. Patients received RUBRACA® 600 mg orally twice daily and concomitant GnRH analog or had prior bilateral orchiectomy. Treatment was continued until disease progression or unacceptable toxicity. The median patient age was 72 years, majority of patients had an ECOG performance status of 0 or 1, 67% of patients had Gleason score of 8 or more at diagnosis, 68% had bone-only disease and 47% had 10 or more bone lesions. The Primary endpoint was Objective Response Rate (ORR) by blinded IRR (Independent Radiology Review), as well as ORR by investigator assessment. Secondary end points included Duration of Response (DOR) in those with measurable disease, locally assessed PSA response rate (50% or more decrease from baseline) rate, Overall Survival (OS), and Safety. The median follow up was 17.1 months.
The confirmed ORR for the IRR-evaluable population was 43.5%, and the confirmed ORR for the investigator-evaluable population was 50.8%. The median DOR was not evaluable and 56% of patients with confirmed Objective Responses had a DOR of 6 months or more. The confirmed PSA response rate was 54.8% and the median time to PSA response was 1.9 months. The Objective Response Rates were similar for patients with a Germline or Somatic BRCA mutations, and for patients with a BRCA1 or BRCA2 mutations. However, a higher PSA response rate was observed in patients with a BRCA2 mutation. The median radiographic Progression Free Survival was 9.0 months per IRR assessment and 8.5 months per investigator assessment. The OS data were not yet mature at the time of the analysis. The most frequent Grade 3 or more treatment related Adverse Event was anemia (25.2%).
It was concluded that RUBRACA® demonstrates promising efficacy in patients with mCRPC with deleterious BRCA mutations. TRITON3 study is evaluating RUBRACA® versus physician’s choice of second-line AR-directed therapy or Docetaxel, in chemotherapy-naïve patients with mCRPC and alterations in BRCA1/2, who progressed on one prior AR-directed therapy.
Rucaparib in Men With Metastatic Castration-Resistant Prostate Cancer Harboring a BRCA1 or BRCA2 Gene Alteration. Abida W, Patnaik A, Campbell D, et al. on behalf of the TRITON2 investigators. J Clin Oncol. 2020;38:3763-3772.

