SUMMARY: Screening both healthy and high-risk populations with a peripheral blood sample (liquid biopsy) has the potential to detect cancer at an early stage, with an increased opportunity to offer curative therapies. Screening assays for cancer should be highly specific with a low rate of false-positive results and overdiagnosis. Analysis of cell-free DNA (cfDNA) with a Liquid Biopsy is presently approved to select EGFR targeted therapies (cobas EGFR mutation test), in patients with advanced Non Small Cell Lung Cancer. However, the role of cell-free DNA analysis for early detection of cancer is not well established.
The Cancer Genome Atlas (TCGA), a landmark cancer genomics program, is a joint effort between the National Cancer Institute and the National Human Genome Research Institute. This program began in 2006 and has molecularly characterized over 20,000 primary cancers and matched normal samples, across 33 different cancer types. After 12 years and contributions from over 11,000 patients, TCGA has deepened our understanding of the molecular basis of cancer, changed the way cancer patients are managed in the clinic, established a rich genomics data resource for the research community and helped advance health and science technologies.
The Circulating Cell-Free Genome Atlas (CCGA) is a prospective, multi-center, observational study and is the largest study ever initiated, to develop a noninvasive, liquid biopsy assay for early cancer detection, based on cell-free DNA (cfDNA). This study completed enrollment of approximately 15,000 participants with and without cancer (56% with 20 tumor types and all clinical stages), across 142 sites in the US and Canada. The principal goal is to develop a noninvasive cancer detection assay and the CCGA was designed to characterize the landscape of genomic cancer signals in the blood and to detect and validate GRAIL’s multi-cancer early detection blood test through three pre-planned sub-studies. The authors in 2018 previously reported that it is possible to detect early-stage lung cancer, with a high degree of specificity, from a simple blood test, using targeted sequencing and whole-genome sequencing. In this substudy, liquid biopsy could accurately detect over 40% of early-stage lung cancers (Stage I-IIIA), with 98% specificity. It was determined that whole-genome bisulfite sequencing for DNA methylation was the most effective approach for early cancer detection. 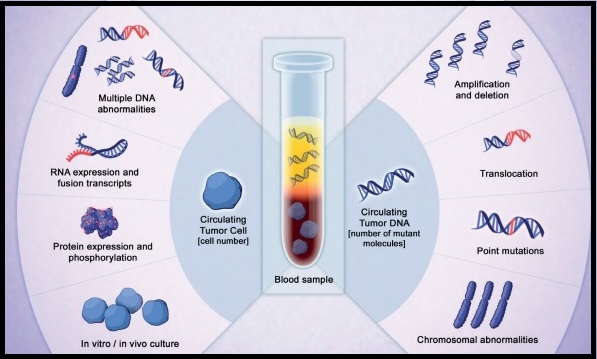
DNA methylation is a natural epigenetic mechanism used by cells to regulate gene expression with some regions of hypermethylation and some regions of hypomethylation, and is a chemical modification to DNA. In cancer, abnormal methylation patterns and the resulting changes in gene expression can contribute to tumor growth (hypermethylation can cause tumor-suppressor genes to be inactivated). Methylation patterns, are unique to the tumor DNA, enabling tumor detection and localization but are not of value when it comes to precision therapies. This is unlike mutations and copy number changes, which can be detected in white blood cells in individuals without cancer as well, leading to false-positives.
In two separate presentations, the authors in this present sub-study reported the results for patients with more than 20 cancer subtypes across all stages and evaluated the prognostic significance of detecting abnormal patterns of cfDNA methylation by whole-genome bisulfite sequencing (WGBS) assay. The goal of targeted methylation assay was to detect both early and advanced disease cancers, and improve clinical outcomes
Liu, MC, et al. reported outcomes for 2,301 participants (1422 had cancer and 879 did not) with more than 20 cancer types (12 prespecified and high-risk cancers included Lung, HR negative Breast, Colorectal, Anorectal, Esophageal, Gastric, Liver, Pancreatic, Head and Neck, Ovary, Myeloma and Lymphoid neoplasms) across all stages. The 12 prespecified cancers account for two thirds of all cancer deaths in the US. At 99% specificity, the sensitivity for these 12 high-risk cancers ranged from 59-86% at early stages (stages I–III). For all 20 cancer types, the overall detection rate across all stages was 55%. Additionally, a Tissue of Origin result was provided for 94% of all cancers detected and of these, the assay correctly identified the Tissue of Origin in 90% of cases, which the authors commented is critical for guiding efficient downstream workup for a positive signal.
Oxnard GR, et al. performed an exploratory longitudinal analysis and reported the results of the Overall Survival of 1,320 participants with more than 20 cancer types in this substudy, thereby evaluating the prognostic significance of detection by this assay. Across all stages of disease, cancers detected by cfDNA whole-genome bisulfite sequencing for DNA methylation were associated with significantly worse survival than those not detected by the blood test. The 2-year Overall Survival was less than 50% among patients whose cancers were detected by the assay compared with 2-year OS of over 90% for those whose cancers were not detected by this assay. The poor prognostic ability of this assay was seen in both cancers that presented with symptoms and those found via screening suggesting that DNA–based detection with this methylation assay may be an indicator of prognosis. In multivariate analysis, cancers detected by this assay had double the risk of death (HR=2.6; P< 0.001) when accounting for clinical stage, cancer type, histologic grade, age, sex, and method of diagnosis and also had comparable prognostic significance to clinical stage (P <0.001).
It was concluded from these two presentations that cfDNA test based on the presence of DNA methylation is highly specific at detecting high-risk malignancies, with very high accuracy for identifying the tissue of origin, and may also have prognostic value.
Genome-wide Cell-free DNA (cfDNA) Methylation Signatures and Effect on Tissue of Origin (TOO) Performance. Liu MC, Jamshidi A, Venn O, et al. 2019 ASCO Annual Meeting. Abstract 3049. Presented June 1, 2019.
Prognostic significance of blood-based cancer detection in plasma cell-free DNA (cfDNA): Evaluating risk of overdiagnosis. Oxnard GR, Chen X, Fung ET, et al. 2019 ASCO Annual Meeting. Abstract 1545. Presented June 3, 2019.

