SUMMARY: Multiple Myeloma is a clonal disorder of plasma cells in the bone marrow and the American Cancer Society estimates that in the United States, about 30,280 new cases will be diagnosed in 2017 and 12,590 patients will die of the disease. Multiple Myeloma is a disease of the elderly, with a median age at diagnosis of 69 years and characterized by intrinsic clonal heterogeneity. With a record number of regulatory approvals for Myeloma treatment over the past 12 years, the median survival for patients with Myeloma is over 10 years. The recent new drugs approved for the treatment of relapsed/refractory Multiple Myeloma include a Histone Decetylase inhibitor (FARYDAK®) and 2 monoclonal antibodies, Daratumumab (DARZALEX®) and Elotuzumab (EMPLICITI®).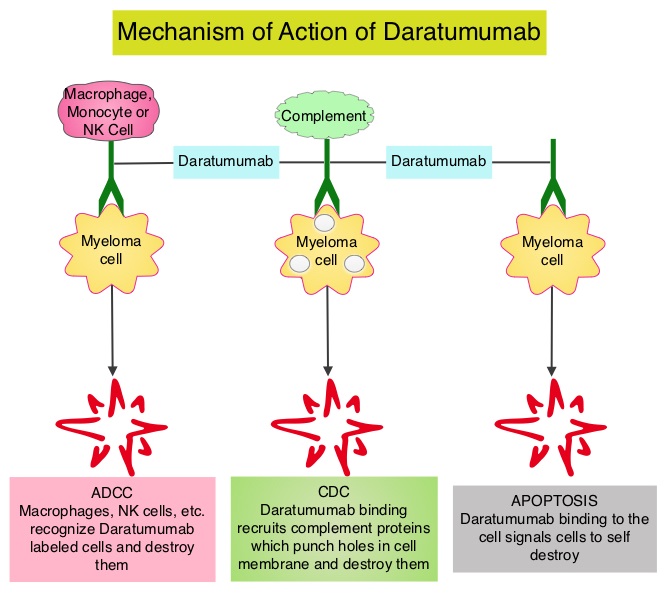
Testing for MRD (Minimal Residual Disease) is standard practice in hematological malignancies such as Chronic Myeloid Leukemia (CML) which is characterized by a defined genetic abnormality ie. Philadelphia Chromosome, and CML also has very effective therapies. Unlike CML, multiple myeloma is a heterogeneous disorder and very effective therapies are only now becoming available. Effective combination regimens for myeloma has resulted in high response rates and therefore achievement of MRD negativity may be a primary endpoint in the near future. MRD negativity has been associated with prolonged Progression Free Survival (PFS) and Overall Survival (OS), in several meta-analyses.
Daratumumab (DARZALEX®) is a human IgG1 antibody that targets CD38, a transmembrane glycoprotein abundantly expressed on malignant plasma cells and with low levels of expression on normal lymphoid and myeloid cells. DARZALEX® exerts its cytotoxic effect on myeloma cells by multiple mechanisms, including Antibody Dependent Cellular Cytotoxicity (ADCC), Complement Mediated Cytotoxicity and direct apoptosis. Additionally, DARZALEX® may have a role in immunomodulation by depleting CD38-positive regulator Immune suppressor cells, and thereby expanding T cells, in patients responding to therapy.
The authors in this publication prospectively evaluated Minimal Residual Disease (MRD) status of patients enrolled in two large phase III trials, the POLLUX and CASTOR studies, and assessed the ability of DARZALEX® to yield deep clinical responses beyond complete remission. In the POLLUX study, 569 patients with relapsed or refractory multiple myeloma were randomized in a 1:1 ratio to receive either DARZALEX®, REVLIMID® (Lenalidomide) and Dexamethasone or REVLIMID® and Dexamethasone. In the CASTOR study, 498 patients with relapsed or refractory multiple myeloma were randomized in a 1:1 ratio to receive either DARZALEX®, VELCADE® (Bortezomib) and Dexamethasone or VELCADE® and Dexamethasone. In both these studies, the addition of DARZALEX® resulted in significant improvements in median PFS (HR=0.37; P<0.001 in the POLLUX study and HR=0.39; P<0.0001) in the CASTOR study), compared to the control group.
The researchers in this study, assessed MRD of bone marrow aspirate samples using ClonoSEQ next-generation sequencing–based assay. In the POLLUX study, MRD was assessed at the time of suspected Complete Response (CR), and at 3 and 6 months after. In the CASTOR study, MRD was assessed at the time of suspected CR, and at 6 months and 12 months after the first dose. The MRD sensitivity thresholds were 0.01% (1 cancer cell per 10,000 nucleated cells, or 10-4), 0.001% (10-5), and 0.0001% (10-6). The MRD negativity rate was defined as the proportion of patients with negative MRD results at any point during the studies. The median follow-up was 13.5 months for the POLLUX study and 7.4 months for the CASTOR study.
In the POLLUX study, the addition of DARZALEX® to REVLIMID® and Dexamethasone improved the MRD-negative status rates from 8.8% to 31.8% at the 10-4 threshold, from 5.7% to 24.8% at the 10-5 threshold, and from 2.5% to 11.9% at the 10-6 threshold. In the CASTOR study, the addition of DARZALEX® to VELCADE® and Dexamethasone improved the MRD-negative status rates from 3.6% to 18.3% at the 10-4 threshold, from 2.4% to 10.4% at the 10-5 threshold, and from 0.8% to 4.4% at the 10-6 threshold. It was noted that the MRD negativity was consistently higher in patients treated with DARZALEX® and DARZALEX® induced MRD negativity in three times as many patients as those treated with standard regimens. Further, MRD negativity was noted as soon as 3 months, with many patients continuing to achieve MRD negativity over time. High risk patients {t(4;14), t(14;16), del17p} benefited as well, with 18% of high risk patients in the POLLUX study and 14% of those in the CASTOR study achieving MRD negativity. Patients with sustained MRD negativity following treatment with DARZALEX®, had a significantly longer PFS compared with the control group.
It was concluded that the addition of DARZALEX®, in relapsed/refractory multiple myeloma, to standard treatment regimens, induced MRD negativity in three times as many patients as those treated with standard regimens. Evaluation of minimal residual disease (MRD) in relapsed/refractory multiple myeloma (RRMM) patients treated with daratumumab in combination with lenalidomide plus dexamethasone or bortezomib plus dexamethasone. Avet-Loisseau H, Casneuf T, Chiu C, et al. Presented at: American Society of Hematology 58th Annual Meeting; December 3-6, 2016; San Diego, CA. Abstract 246

