
Written by: Thomas E Boyd, MD, Texas Oncology
Content Sponsored by: Bristol Myers Squibb
Dr. Boyd is a paid consultant for BMS and was compensated for his contribution in drafting this article.
Acute myeloid leukemia (AML) is an aggressive hematologic cancer with 5-year relative survival rates less than 30%.1 The journey for patients diagnosed with AML is very complex, and helping patients achieve remission is a major goal of initial treatment.2 Relapse after this initial therapy poses a significant roadblock to survival, as evidenced by high relapse rates and the current 5-year survival rate.1,3 There remains a critical need for therapies that extend survival.4
After a patient enters first remission, continued AML treatment is one avenue for appropriate patients.4 ONUREG® is the first and only FDA-approved continued AML treatment for adult patients with acute myeloid leukemia who achieved first complete remission (CR) or complete remission with incomplete blood count recovery (CRi) following intensive induction chemotherapy and are not able to complete intensive curative therapy.5
The efficacy and safety of ONUREG® was evaluated in the Phase 3 QUAZAR®AML-001 trial, where patients who completed intensive induction with or without consolidation therapy were randomized 1:1 to receive ONUREG or placebo orally on Days 1 through 14 of each 28-day cycle.5 Randomization was stratified by age at time of induction therapy, cytogenetic risk category at time of induction therapy, prior history of MDS/CMML, and receipt of consolidation therapy following induction therapy.5
Median OS, the primary endpoint in the QUAZAR®AML-001 trial, was over 2 years (24.7 months) in the ONUREG® arm and 14.8 months in the placebo arm (hazard ratio [HR] 0.69, 95% confidence interval [CI]: 0.55-0.86; P=0.0009; Figure 1).5 This statistically significant survival benefit of ~10 months for patients with AML in first remission who received ONUREG® compared to placebo demonstrated that QUAZAR® AML-001 trial met its primary endpoint.5 Survival estimates were 73% for ONUREG® (n=168; 95% CI: 67, 78) and 56% for placebo (n=127; 95% CI: 49, 62) at 1 year, and 51% for ONUREG® (n=115; 95% CI: 44, 57) and 37% for placebo (n=82; 95% CI: 31, 43) at 2 years.6 Analyses of survival estimates at 1 and 2 years were not designed to show a difference between treatment arms.
Additional analyses show the influence of ONUREG® across subgroups vs placebo for median OS (Figure 2). It is important to note that these prespecified analyses should be interpreted with caution because of potential selection bias, insufficient sample size, and a higher probability of making a false positive finding.7
The most common adverse reactions (ARs, ≥ 10%) associated with ONUREG® treatment included nausea, vomiting, diarrhea, fatigue/asthenia, constipation, pneumonia, abdominal pain, arthralgia, decreased appetite, febrile neutropenia, dizziness, and pain in extremity.5 Serious ARs occurred in 15% of patients who received ONUREG®, and the most common Grade 3/4 ARs were febrile neutropenia (11%), pneumonia (9%), diarrhea (5%), and fatigue/asthenia (4%).5
Efficacy and safety were established by the large, multicenter QUAZAR® AML-001 trial. These results established ONUREG® as a continued treatment for adult patients with AML who achieve first complete remission (CR) or complete remission with incomplete blood count recovery (CRi) following intensive induction chemotherapy and are not able to complete intensive curative therapy.
Figure 1: Kaplan-Meier curve for OS: ITT population in QUAZAR® AML-0015
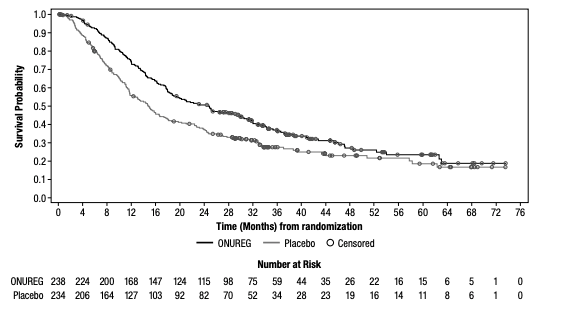
Figure 2: Overall survival select subgroup analysis6,7
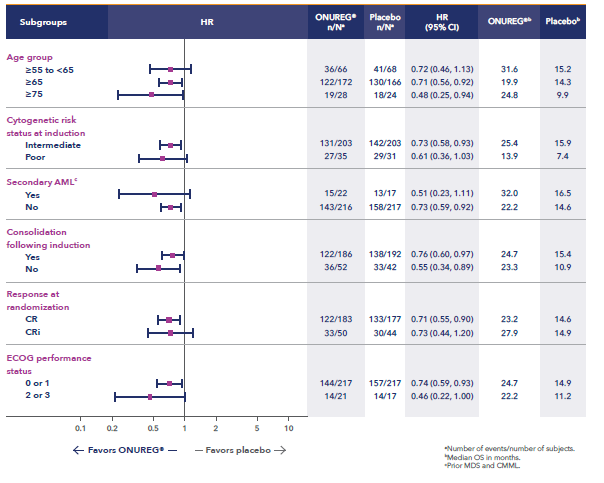
*Analysis limitations: These prespecified subgroup analyses should be interpreted with caution to determine a difference between arms in these select subgroups because of potential selection bias, insufficient sample size, and a higher probability of making a false positive finding.
IMPORTANT SAFETY INFORMATION
CONTRAINDICATIONS
ONUREG® is contraindicated in patients with known severe hypersensitivity to azacitidine or its components.
WARNINGS AND PRECAUTIONS
Risks of Substitution with Other Azacitidine Products
Due to substantial differences in the pharmacokinetic parameters, the recommended dose and schedule for ONUREG® are different from those for the intravenous or subcutaneous azacitidine products. Treatment of patients using intravenous or subcutaneous azacitidine at the recommended dosage of ONUREG® may result in a fatal adverse reaction. Treatment with ONUREG® at the doses recommended for intravenous or subcutaneous azacitidine may not be effective. Do not substitute ONUREG® for intravenous or subcutaneous azacitidine.
Myelosuppression
New or worsening Grade 3 or 4 neutropenia and thrombocytopenia occurred in 49% and 22% of patients who received ONUREG®. Febrile neutropenia occurred in 12%. A dose reduction was required for 7% and 2% of patients due to neutropenia and thrombocytopenia. Less than 1% of patients discontinued ONUREG® due to either neutropenia or thrombocytopenia. Monitor complete blood counts and modify the dosage as recommended. Provide standard supportive care, including hematopoietic growth factors, if myelosuppression occurs.
Increased Early Mortality in Patients with Myelodysplastic Syndromes (MDS)
In AZA-MDS-003, 216 patients with red blood cell transfusion-dependent anemia and thrombocytopenia due to MDS were randomized to ONUREG® or placebo. 107 received a median of 5 cycles of ONUREG® 300 mg daily for 21 days of a 28-day cycle. Enrollment was discontinued early due to a higher incidence of early fatal and/or serious adverse reactions in the ONUREG® arm compared with placebo. The most frequent fatal adverse reaction was sepsis. Safety and effectiveness of ONUREG® for MDS have not been established. Treatment of MDS with ONUREG® is not recommended outside of controlled trials.
Embryo-Fetal Toxicity
ONUREG® can cause fetal harm when administered to a pregnant woman. Azacitidine caused fetal death and anomalies in pregnant rats via a single intraperitoneal dose less than the recommended human daily dose of oral azacitidine on a mg/m2 basis. Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with ONUREG® and for at least 6 months after the last dose. Advise males with female partners of reproductive potential to use effective contraception during treatment with ONUREG® and for at least 3 months after the last dose.
ADVERSE REACTIONS
Serious adverse reactions occurred in 15% of patients who received ONUREG®. Serious adverse reactions in ≥2% included pneumonia (8%) and febrile neutropenia (7%). One fatal adverse reaction (sepsis) occurred in a patient who received ONUREG®.
Most common (≥10%) adverse reactions with ONUREG® vs placebo were nausea (65%, 24%), vomiting (60%, 10%), diarrhea (50%, 21%), fatigue/asthenia (44%, 25%), constipation (39%, 24%), pneumonia (27%, 17%), abdominal pain (22%, 13%), arthralgia (14%, 10%), decreased appetite (13%, 6%), febrile neutropenia (12%, 8%), dizziness (11%, 9%), pain in extremity (11%, 5%).
LACTATION
There are no data regarding the presence of azacitidine in human milk or the effects on the breastfed child or milk production. Because of the potential for serious adverse reactions in the breastfed child, advise women not to breastfeed during treatment with ONUREG® and for 1 week after the last dose.
Please see full Prescribing Information for ONUREG®.
References
1. National Cancer Institute. SEER Cancer Statistics Factsheets: Acute Myeloid Leukemia. http://seer.cancer.gov/statfacts/html/amyl.html. Accessed April 21, 2021.
2. Medeiros BC. Interpretation of clinical endpoints in trials of acute myeloid leukemia. Leuk Res. 2018;68:32-29.
3. Wei AH. Maintenance therapy for AML: are we there yet? Blood. 2019;133(13):1390-1392.
4. Medeiros BC, Chan SM, Daver NG, Jonas BA, Pollyea DA. Optimizing survival outcomes with post-remission therapy in acute myeloid leukemia. Am J Hematol. 2019;94:803-811.
5. ONUREG® [Prescribing Information]. Summit, NJ: Celgene Corporation; 2021.
6. Wei AH, Döhner H, Pocock C, et al. The QUAZAR AML-001 maintenance trial: results of a phase III international, randomized, double-blind, placebo-controlled study of CC-486 in patients with acute myeloid leukemia (AML) in first remission [oral presentation at ASH 2019]. Blood. 2019;134(Suppl 2):LBA-3.
7. Wei AH, Döhner H, Pocock C, et al. Oral Azacitidine Maintenance Therapy for Acute Myeloid Leukemia in First Remission. N Engl J Med. 2020;383:2526-2537.
© 2021 Celgene Corporation.
ONUREG is a trademark of Celgene Corporation, a Bristol Myers Squibb company.
QUAZAR® is a registered trademark of Celgene Corporation, a Bristol Myers Squibb company
02/22 2011-US-2100200

