SUMMARY: The FDA on July 17, 2017 approved NERLYNX® (Neratinib) for the extended adjuvant treatment of adult patients with early stage HER2-overexpressed/amplified breast cancer, to follow adjuvant Trastuzumab (HERCEPTIN®)-based therapy. Breast cancer is the most common cancer among women in the US and about 1 in 8 women (12%) will develop invasive breast cancer during their lifetime. Approximately, 255,180 new cases of invasive breast cancer will be diagnosed in 2017 and over 41,070 women will die of the disease. The HER or erbB family of receptors consist of HER1, HER2, HER3 and HER4. Approximately 15%-20% of invasive breast cancers overexpress HER2/neu oncogene, which is a negative predictor of outcomes without systemic therapy. HERCEPTIN® (Trastuzumab) is a humanized monoclonal antibody targeting HER2 and adjuvant chemotherapy given along with HERCEPTIN® reduces the risk of disease recurrence and death, among patients with HER2-positive, early breast cancer. Nonetheless, approximately 25% of patients will develop recurrent disease within 10 years following this adjuvant intervention. Extending the duration of adjuvant HERCEPTIN® therapy or adding TYKERB® (Lapatinib), a Tyrosine Kinase Inhibitor that targets HER1 and HER2, has not improved outcomes.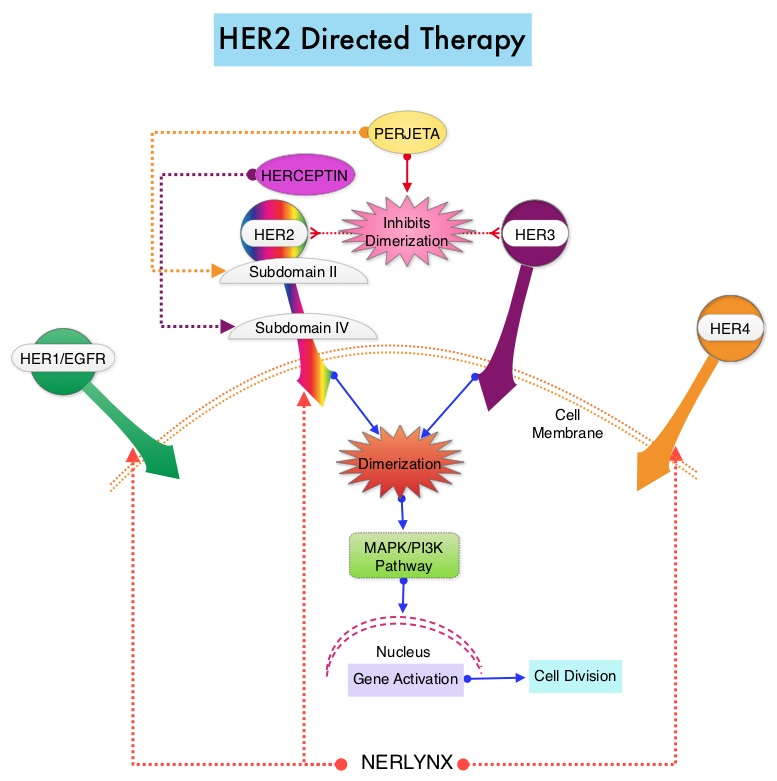
NERLYNX® is a potent, irreversible, oral Tyrosine Kinase Inhibitor, of HER1, HER2 and HER4 (pan-HER inhibitor). NERLYNX® interacts with the catalytic domain of HER1, HER2, and HER4 and blocks their downstream signaling pathways, resulting in decreased cell proliferation and increased cell death. Clinical data has suggested that NERLYNX® has significant activity in suppressing HER-mediated tumor growth and is able to overcome tumor escape mechanisms experienced with current HER2-targeted and chemotherapeutic agents. It has been well known that hormone receptor positive breast cancer patients, who are also HER2-positive, have relative resistance to hormone therapy. Preclinical models had suggested that the addition of NERLYNX® could improve responses in ER positive, HER2-positive breast cancer patients. Further, NERLYNX® has clinical activity in patients with HER2-positive metastatic breast cancer.
The approval of NERLYNX® was based on ExteNET trial, which is a multicentre, randomized, double-blind, placebo-controlled, phase III study, in which the efficacy and safety of 12 months of NERLYNX® after HERCEPTIN®-based adjuvant therapy was evaluated, in patients with early stage HER2-positive breast cancer. Patients with early stage HER2-positive breast cancer (N=2,840), and within two years of completing adjuvant HERCEPTIN®, were randomized in a 1:1 ratio to receive either oral NERLYNX® 240 mg per day (N=1420) or placebo (N=1420), for one year. Patients were stratified by hormone receptor status, nodal status (0, 1-3, or 4 or more), and HERCEPTIN® adjuvant regimen (sequentially versus concurrently with chemotherapy). The Primary endpoint was invasive Disease Free Survival (iDFS), defined as the time between the randomization date to the first occurrence of invasive recurrence (local/regional, ipsilateral or contralateral breast cancer), distant recurrence, or death from any cause, within two years of follow up. The median follow up was 2 years.
In the updated analysis, the two year iDFS was 94.2% in patients treated with NERLYNX® compared with 91.9% in those receiving placebo (HR 0.66; P=0.008). Patients with ER positive breast cancer were noted to have greater benefit. The most common grade 3-4 adverse events associated with NERLYNX® were diarrhea, vomiting and nausea. Patients can experience diarrhea early, in the first 2 or 3 days and this can be alleviated using antidiarrheal prophylaxis with Loperamide, initiated with the first dose of NERLYNX® and continued for the first 2 months of treatment and as needed thereafter.
It was concluded that NERLYNX® when given for 12 months after chemotherapy and HERCEPTIN®-based adjuvant therapy, to women with HER2-positive breast cancer, significantly improved 2-year invasive Disease Free Survival. Longer follow up will hopefully address if there is an Overall Survival benefit with this treatment intervention. NERLYNX® is the first TKI approved by the FDA, shown to reduce the risk for disease recurrence, in patients with early stage HER2-positive breast cancer. Neratinib after trastuzumab-based adjuvant therapy in patients with HER2-positive breast cancer (ExteNET): a multicentre, randomised, double-blind, placebo-controlled, phase 3 trial Chan A, Delaloge S, Holmes FA, et al. The Lancet Oncology 2016; 17:367- 377

