SUMMARY: The US Food and Drug Administration on January 29, 2015, approved Ibrutinib (IMBRUVICA®) for the treatment of patients with Waldenstrom Macroglobulinemia (WM). Waldenstrom Macroglobulinemia is classified as a LymphoPlasmacytic Lymphoma (LPL) according to the Revised European American Lymphoma (REAL) and WHO (World Health Organization) classifications. It is estimated that about 1500 new cases of WM will be diagnosed each year in the US. Chromosome 6q deletion detected by FISH technique on bone marrow evaluation, is seen in 40-50% of the patients with WM. Whole genome sequencing of lymphoplasmacytic cells has revealed activating somatic mutations in MYD88 (L265P) and CXCR4 genes, in Waldenstrom Macroglobulinemia (WM).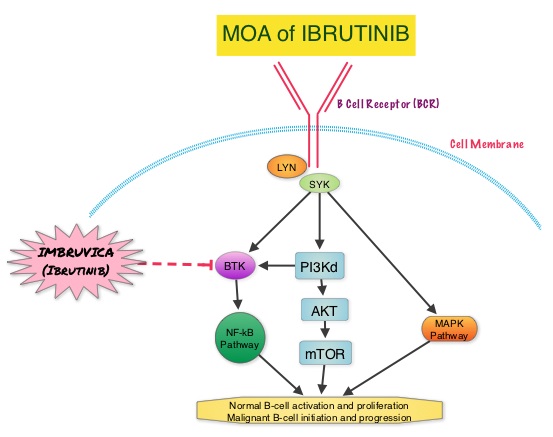 About 90% of patients with Waldenstrom Macroglobulinemia demonstrate a mutation in chromosome 3p (MYD88 L265P), which is specific to WM and may be an early oncogenic event in WM pathogenesis.. It appears that MYD88 L265P promotes malignant cell proliferation via the Bruton’s Tyrosine Kinase (BTK) signaling pathway. Mutations in CXCR4 gene are present in 30% of patients with WM, and their expression induces BTK activity and may confer resistance to BTK inhibitors. IMBRUVICA® (Ibrutinib) is an oral, irreversible inhibitor of BTK and inhibits cell proliferation and promotes programmed cell death (Apoptosis). Preliminary studies in WM patients have revealed that IMBRUVICA® prevents the binding of MYD88 L265P (mutated gene) to BTK thereby selectively killing tumor cells. On the other hand it was noted that a major response to IMBRUVICA® was less likely when mutations in CXCR4 gene were present in the tumor cells. With this molecular understanding of WM, the authors enrolled 63 patients with relapsed/refractory symptomatic WM and were treated with IMBRUVICA®, 420 mg PO daily for 2 years or until disease progression or unacceptable toxicity. Anemia was main indication for treatment initiation (87.3% of patients) and the mean baseline hemoglobin level was 10.5 g/dL, mean serum IgM level was 3610 mg/dL, 70% had bone marrow involvement and 60% of patients had lymphadenopathy.
About 90% of patients with Waldenstrom Macroglobulinemia demonstrate a mutation in chromosome 3p (MYD88 L265P), which is specific to WM and may be an early oncogenic event in WM pathogenesis.. It appears that MYD88 L265P promotes malignant cell proliferation via the Bruton’s Tyrosine Kinase (BTK) signaling pathway. Mutations in CXCR4 gene are present in 30% of patients with WM, and their expression induces BTK activity and may confer resistance to BTK inhibitors. IMBRUVICA® (Ibrutinib) is an oral, irreversible inhibitor of BTK and inhibits cell proliferation and promotes programmed cell death (Apoptosis). Preliminary studies in WM patients have revealed that IMBRUVICA® prevents the binding of MYD88 L265P (mutated gene) to BTK thereby selectively killing tumor cells. On the other hand it was noted that a major response to IMBRUVICA® was less likely when mutations in CXCR4 gene were present in the tumor cells. With this molecular understanding of WM, the authors enrolled 63 patients with relapsed/refractory symptomatic WM and were treated with IMBRUVICA®, 420 mg PO daily for 2 years or until disease progression or unacceptable toxicity. Anemia was main indication for treatment initiation (87.3% of patients) and the mean baseline hemoglobin level was 10.5 g/dL, mean serum IgM level was 3610 mg/dL, 70% had bone marrow involvement and 60% of patients had lymphadenopathy.  Sanger sequencing was used to determine MYD88 and CXCR4 mutations in the bone marrow lymphoplasmacytic cells. At best response, the median serum IgM levels declined to 1340 mg/dL (P<0.00001), median hemoglobin rose to 12.6 g/dL, (P<0.00001). At 6 months, bone marrow assessment post treatment, demonstrated a reduction in WM disease involvement from 70% to 45% (P=0.0006). With a median follow up at 6 cycles, the best overall response rate was 81% and a median time to response was 4 weeks. In patients who underwent tumor sequencing, mutations in CXCR4 gene impacted response rates. The major response rate for patients with wild-type CXCR4 gene was 77% compares to 30% in those with CXCR4 mutations (p=0.018). Further, patients with wild-type CXCR4 also had increased peripheral lymphocytosis following treatment with IMBRUVICA® compared to those with CXCR4 mutations (P=0.001). The most common more than grade 2 treatment related toxicities included thrombocytopenia (14.3%) and neutropenia (19.1%). The authors concluded that IMBRUVICA® is highly active in patients with relapsed or refractory Waldenstrom Macroglobulinemia, with rapid reductions in serum IgM level and improved hemoglobin levels. The presence of CXCR4 mutations negatively impact response rates in this patient group. Treon SP, Tripsas CK, Yang G, et al. Presented at: 55th ASH Annual Meeting; December 7-10, 2013; New Orleans, LA. Abstract 251.
Sanger sequencing was used to determine MYD88 and CXCR4 mutations in the bone marrow lymphoplasmacytic cells. At best response, the median serum IgM levels declined to 1340 mg/dL (P<0.00001), median hemoglobin rose to 12.6 g/dL, (P<0.00001). At 6 months, bone marrow assessment post treatment, demonstrated a reduction in WM disease involvement from 70% to 45% (P=0.0006). With a median follow up at 6 cycles, the best overall response rate was 81% and a median time to response was 4 weeks. In patients who underwent tumor sequencing, mutations in CXCR4 gene impacted response rates. The major response rate for patients with wild-type CXCR4 gene was 77% compares to 30% in those with CXCR4 mutations (p=0.018). Further, patients with wild-type CXCR4 also had increased peripheral lymphocytosis following treatment with IMBRUVICA® compared to those with CXCR4 mutations (P=0.001). The most common more than grade 2 treatment related toxicities included thrombocytopenia (14.3%) and neutropenia (19.1%). The authors concluded that IMBRUVICA® is highly active in patients with relapsed or refractory Waldenstrom Macroglobulinemia, with rapid reductions in serum IgM level and improved hemoglobin levels. The presence of CXCR4 mutations negatively impact response rates in this patient group. Treon SP, Tripsas CK, Yang G, et al. Presented at: 55th ASH Annual Meeting; December 7-10, 2013; New Orleans, LA. Abstract 251.

